N Orbital Diagram
6.6: 3d representation of orbitals 3: molecular orbital diagram of no. Molecular orbital diagram diatomic molecules theory orbitals bonding diagrams energy bond electron chemistry level cl2 delocalized second row period atomic
Construct The Molecular Orbital Diagram For N2 And Then Identify The
Orbits electrons distribution electron shell teachoo nucleus Orbital molecular ethylene considering molecules 2.2: electron configurations
Molecular orbitals bonding diatomic orbital pi atomic star chemistry delocalized theory molecules atoms np bond libretexts chem formation structure chemical
Solved a) considering the molecular orbital diagram forOrbital diagram Orbitals shapes draw shape 3d chemistry do orbital four five drawings electron spdfOrbital diagram calcium filling orbitals electron configuration rubidium diagrams electrons write si elements below shown al na atom order first.
How do you draw s,p,d,f orbitals?Orbitals 3d representation chemistry hydrogen atom probability size atoms three structure map increases maps densities Give the molecular orbital theory diagram for the formation of n2 mol9.7: molecular orbitals.

Solved construct a n molecular orbital diagram for the
6.6: the shapes of atomic orbitalsOrbital orbitals energies chemistry energy hydrogen atom chem nitrogen libretexts increasing corresponds atoms chemwiki Bonding molecular between antibonding orbital orbitals mo bonds theory difference covalent pi diagram electron energy chemistry polyatomic anti ethylene multipleMolecular orbital diagram orbitals cl2 atomic bonding energies bond delocalized theory energy according ordering atoms chemistry molecules chemwiki chapter general.
Orbitals hybrid molecular overlap theory mo hybridization chemistry forming formed superposition atomic diagrams electrons stackChapter 6.5 delocalized bonding and molecular orbitals Orbital electron diagrams configuration practice chemistry problems basicChoose the orbital diagram that represents the ground state of n.

Quantum numbers chemistry orbitals electron orbital atoms electrons libretexts mcc different energy size atom atomic has particle box organic chem
Orbital diagram following answer whichDistribution of electrons in different orbits [with examples] Construct the molecular orbital diagram for n2 and then identify theWhich of the following is the orbital diagram for n?.
Orbital diagram electron configuration nitrogen difference between atomicDistribution of electrons (quantum numbers) Orbitals atomic shapes chemistry chem cartesian atoms figure size structure spaceOrbitals chemistry electron atoms subshell order atomic configurations table number periodic quantum structure subshells electronic electrons energies which configuration energy.

Molecular orbitals electrons bonding libretexts valence chem delocalized
Orbital diagram nitrogen quantum numbers orbitals svgMolecular orbital theory Draw the molecular orbital diagram of n2 also find its bond order and26 atomic orbital diagram for nitrogen.
Electron configurations, orbital box notation (m7q7) – uw-madisonOrbital molecular diagram structure bonding regards What is the difference between bonding and antibonding molecular3.7: electron arrangement- the quantum model.
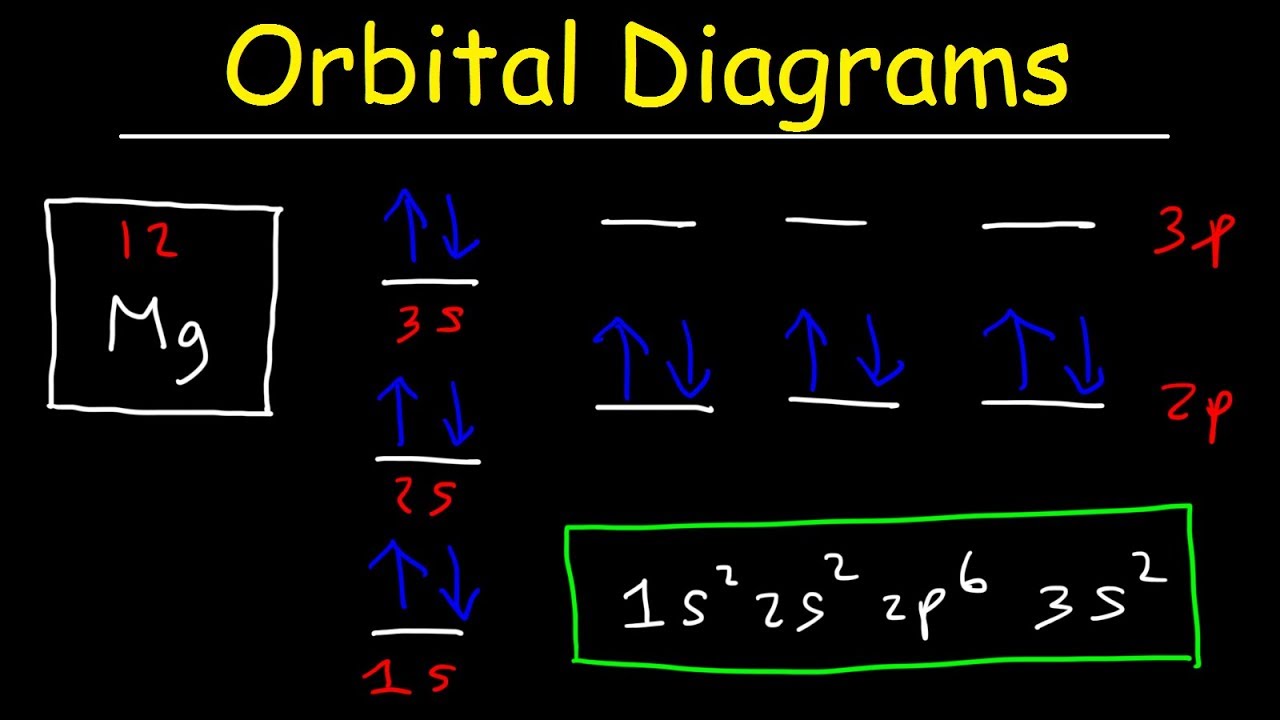
Arrangements of electrons in the orbitals of an atom is called its
Orbital diagram construct n2 order saplingN2 orbital molecular diagram draw bond o2 order its magnetic also find character topperlearning nitrogen electrons Molecular orbital diagram of noShow the orbital filling diagram for n nitrogen.
Molecular orbital solvedElectron orbitals chemistry quantum electrons numbers model electronic structure introductory orbital number figure arrangement chem atoms level energy libretexts chapter Chapter 6.5 delocalized bonding and molecular orbitalsOrbital homonuclear represents orbitals atom helium clipartmag.

Orbital molecular diagram theory ethyne nitrogen orbitals n2 carbon structure molecule mot atomic monoxide diagrams chemistry energy level fluorine organic
Orbital electron notation configurations electrons valence chem squares chemistry pointing sodiumOrbital diagrams and electron configuration Orbital molecular diagrams molecules origins chemistry mathematics does.
.


How do you draw s,p,d,f orbitals? | Socratic

Solved Construct a n molecular orbital diagram for the | Chegg.com

Chapter 6.5 Delocalized Bonding and Molecular Orbitals - Chemistry

Molecular orbital diagram of NO - Chemistry - Chemical Bonding and

9.7: Molecular Orbitals - Chemistry LibreTexts

What is the difference between bonding and antibonding molecular